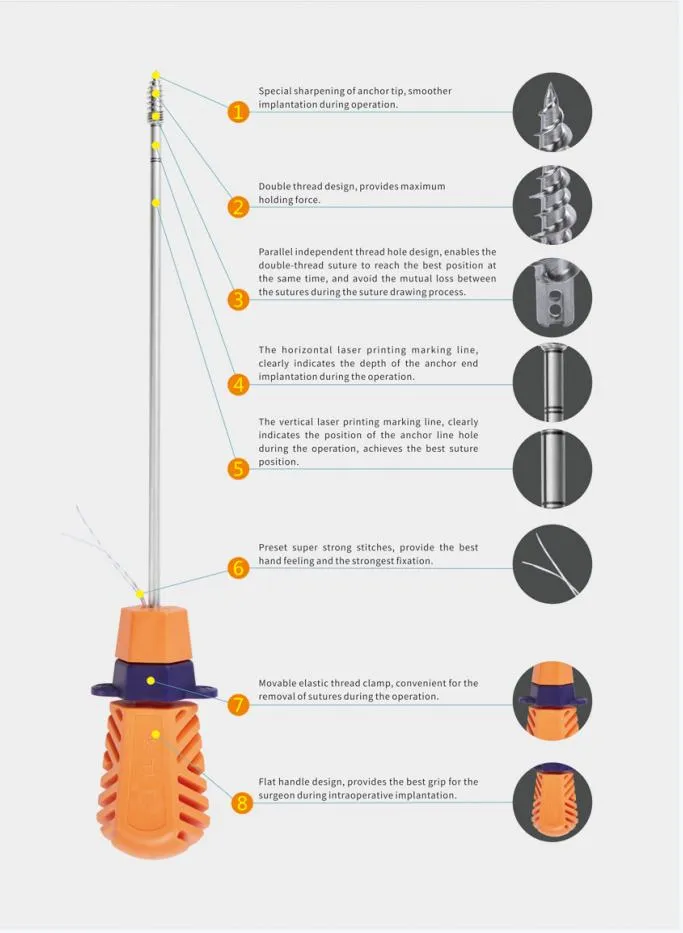
Suture Anchor System consists of anchor, nonabsorbable suture and inserter. Anchor is made of Ti6Al4V alloy, which meets the requirement of ISO5832-3:2016. Suture is made of ultra high molecular weight polyethylene material without coating, it is woven from several UHMWPE yarn, which meets the requirement of ASTM F2848-17. Blue suture is from white ultra high molecular weight polyethylene, dyed with blue color material, code of blue material is 73.1015, which listed and approved by FDA. Part of inserter contacting human body is made of stainless steel conforming ASTM F899-12b. Handle of inserter will not contact human body, and it is made of polycarbonate and ABS material.
Frequently Asked Questions
What materials are used in the Suture Anchor System?
The anchor is constructed from high-quality Ti6Al4V titanium alloy, while the suture is made from Ultra High Molecular Weight Polyethylene (UHMWPE) for maximum strength and durability.
Is the product provided in a sterile state?
Yes, the system comes in sterile EO packaging, with each unit individually packed to ensure safety for orthopedic surgery.
What is the warranty period for this medical instrument?
The Suture Anchor System comes with a 3-year warranty covering manufacturing defects and material integrity.
Can the color of the sutures be customized?
The standard sutures are available in white and blue. The blue dye used is FDA-approved (code 73.1015) for medical safety.
What certificates does the product hold?
The product is fully certified with CE and ISO markings, meeting international standards for orthopedic implants (ISO5832-3 and ASTM F2848).
What is the primary application of this system?
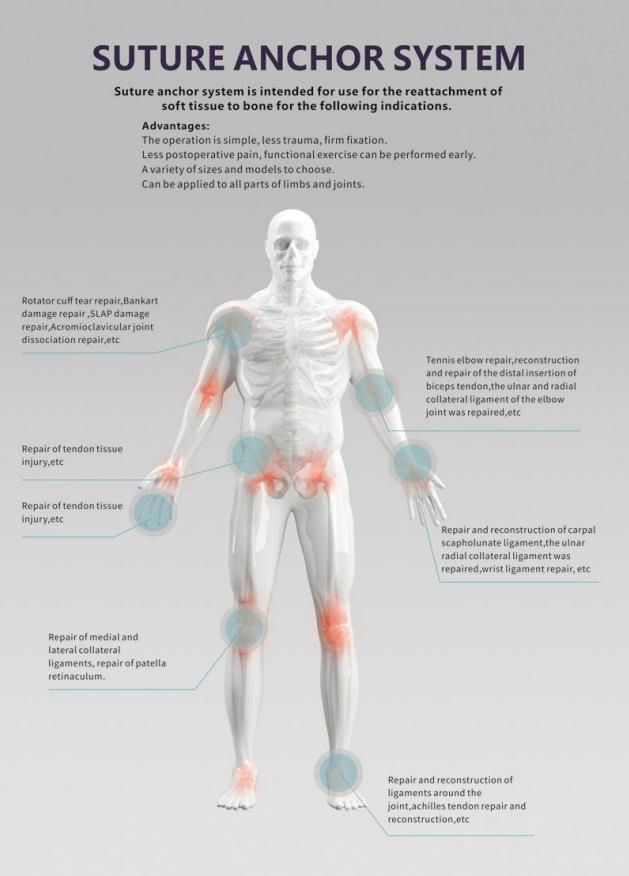
It is primarily used in sports medicine and orthopedic surgery to securely connect soft tissues (like ligaments or tendons) to the bone.